how many electrons in f orbital|For s, p, d, and f orbitals, how many electrons can each hold? : Tagatay When l = 3 (f-type orbitals), m l can have values of –3, –2, –1, 0, +1, +2, +3, and we can have seven 4f orbitals. Thus, we find a total of 16 orbitals in the n = 4 shell of an atom. Play at the best online sportsbook sites in the USA in 2024. Make your dreams come true with real money bets. New sportsbook games online. Start betting!
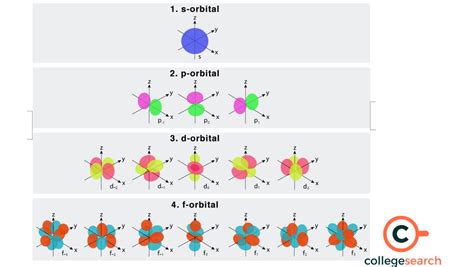
how many electrons in f orbital,This means that the s orbital can contain up to two electrons, the p orbital can contain up to six electrons, the d orbital can contain up to 10 electrons, and the f orbital can contain up to 14 .When l = 3 (f-type orbitals), m l can have values of –3, –2, –1, 0, +1, +2, +3, and we can have seven 4f orbitals. Thus, we find a total of 16 orbitals in the n = 4 shell of an atom. Oxygen (atomic number 8) has a pair of electrons in any one of the 2 p orbitals (the electrons have opposite spins) and a single electron in each of the other two. Fluorine (atomic number 9) has only .For s, p, d, and f orbitals, how many electrons can each hold? Electron shells consist of one or more subshells, and subshells consist of one or more atomic orbitals. Electrons in the same subshell have the same energy, while electrons in different shells or subshells have different energies. Created by Sal Khan.
The f orbital has 7 sub levels with the possibility of two electrons in each suborbital. Therefore, the f orbital can hold 14 electrons.

An orbital is a region around an atom's nucleus where electrons are likely to be found. Different types of orbitals (s, p, d, f) have different shapes and can hold .Learn about the shapes, sizes and capacities of s,p,d,f orbitals, the regions of space where electrons are most likely to be found. Find out how many electrons can s,p,d,f orbitals hold and see examples of electron .

Answer link. 2 electrons ---> see below: The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only . Answer link. 2 electrons ---> see below: The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only .7f atomic orbitals. For any atom, there are seven 7f orbitals. The f-orbitals are unusual in that there are two sets of orbitals in common use.The first set is known as the general set, this page.The second set .
The energy levels are labeled with an n value, where n = 1, 2, 3, .. Generally speaking, the energy of an electron in an atom is greater for greater values of n. This number, n, is referred to as the principal quantum number. Figure 5.7.1 5.7. 1: Different energy levels are numbered by principal quantum numbers n.The largest value of the Principle Quantum Number (n) is 3, so that is the outermost orbital. Counting the number of electrons, we find that only the s orbital is present and it has only one electron. So Na has one electron in its outermost orbital. Another example that I'll use is Fluorine (F). Its electronic configuration is 1s2, 2s2, 2p5.
This tells us that each subshell has double the electrons per orbital. The s subshell has 1 orbital that can hold up to 2 electrons, the p subshell has 3 orbitals that can hold up to 6 electrons, the d subshell has 5 orbitals that hold up to 10 electrons, and the f subshell has 7 orbitals with 14 electrons. An orbital is a region around an atom's nucleus where electrons are likely to be found. Different types of orbitals (s, p, d, f) have different shapes and can hold different numbers of .Figure 9.6.5: Electrons are added to atomic orbitals in order from low energy (bottom of the graph) to high (top of the graph) according to the Aufbau principle. Principle energy levels are color coded, while sublevels are grouped together and each circle represents an orbital capable of holding two electrons.how many electrons in f orbital For s, p, d, and f orbitals, how many electrons can each hold?The four chemically important types of atomic orbital correspond to values of l = 0, 1, 2, and 3. Orbitals with l = 0 are s orbitals and are spherically symmetrical, with the greatest probability of finding the electron occurring at the nucleus. All orbitals with values of n > 1 and l = 0 contain one or more nodes.how many electrons in f orbital 1 Answer. The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only hold up to 2 electrons. 2 electrons ---> see below: The f sublevel as a whole can hold up to 14 electrons due to the fact that it consists of 7 orbitals, but each one can only hold up to 2 electrons.
how many electrons in f orbital|For s, p, d, and f orbitals, how many electrons can each hold?
PH0 · s,p,d,f Orbitals
PH1 · What is the maximum number of electrons an f
PH2 · The periodic table, electron shells, and orbitals
PH3 · Shells, subshells, and orbitals (video)
PH4 · Quantum numbers (video)
PH5 · How many electrons can an f orbital have?
PH6 · For s, p, d, and f orbitals, how many electrons can each hold?
PH7 · Electronic Orbitals
PH8 · 8.3: Electron Configurations
PH9 · 2.2: Atomic Orbitals and Quantum Numbers